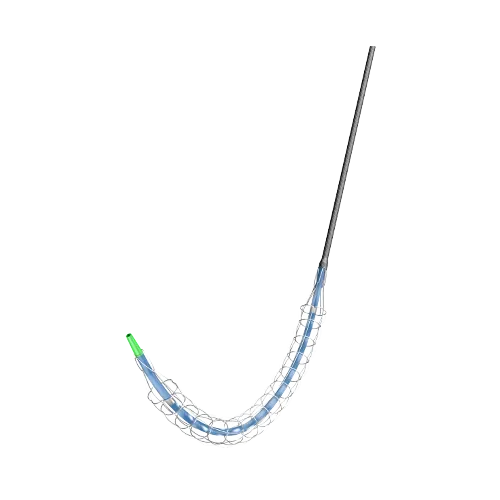
PRODUCTS
Barty Medical mainly develop, manufacture and market vascular interventional devices with independent intellectual property rights, including coronary drug coated balloons, peripheral drug coated balloons, coronary PTCA balloons, peripheral PTA balloons, and catheter sheaths, angiography catheters, guide catheters, etc.
Barty Medical
ABOUT US
Zhejiang Barty Medical Technology Co., Ltd. (“Barty Medical”) is a national high-tech enterprise dedicated to the R&D, manufacturing and sales of medical devices in the field of vascular intervention.
Founded in August 2015, the company is located in Hangzhou City. It has a 1,900 m2 GMP class 10,000 (partially class 100) cleanroom, equipped with professional and complete facilities and equipment.
NEWS CENTER

Brand New Excipient-Free Paclitaxel Nano-Scale Drug-Coated Balloon – Barty Medical FLOWTYTM Paclitaxel-Eluting PTCA Balloon Catheter
As a representative work of implant free concept, Drug coated balloon has received widespread attention
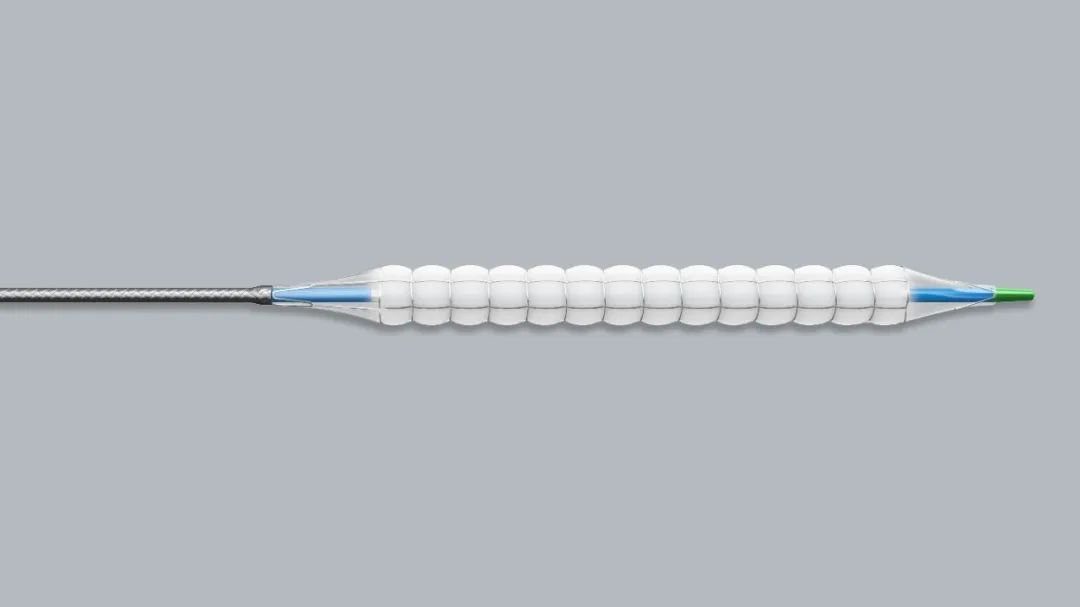
Barty Medical’s nitinol drug coated PTCA balloon was approved for national innovative medical device special green channel
On June 9, 2022, the nitinol drug coated PTCA balloon independently developed by Zhejiang Barty

Secretary Yao Jun of Zhejiang Sci-tech University came to Barty to carry out school-enterprise cooperation
In order to deepen school-enterprise cooperation, on the morning of June 2, 2022, Yao Jun,